 
This molecule also has a linear geometry because the carbon has only two atoms and no lone pairs on it.īoron is the central atom, so we can draw the skeletal structure as: We have seen how the BeCl 2 adopts a linear geometry because of the two Cl atoms on the Be. So, let’s discuss these geometries in a little more detail. To make this easier, we can classify the compounds into two groups: the ones with lone pairs on the central atoms, and the ones without them.įor central atoms with no lone pairs, there are 5 molecular geometries and approximate bond angles you need to know. For example, the oxygen in water has two atoms (hydrogens) connected to it, but when determining is geometry, we need to also consider the effect of the two lone pairs: Now, it is not always that the central atom has no lone pairs, and we only need to worry about the number of atoms. Let’s put a little chart of example for steric number ranging from 2-6: 
So, once again, keep in mind that multiple bonds do not matter – we treat them as single bonds and only count the atoms and lone pairs for the electron group/steric number. Another important thing you need to remember is that it does not matter whether the central atom is connected with single, double, or triple bonds – we only count the number of atoms and lone pairs it bears.įor example, the steric number of carbon in HCN is still two even though it is connected with the nitrogen via a triple bond: 
Notice that we do not count the lone pairs on the Cl atoms, since we are determining the geometry of the central atom. The remaining 12 go on the Cl atoms as lone pairs:īecause the Be has only two atoms and no lone pairs, its steric number is two. There is a total of 2 + 2×7 = 16 valence electrons, and we use four of them to make the bonds. Beryllium is the central atom and, therefore, the structure would be: So, the first step is to draw the correct skeletal structure. * Put more electronegative elements in terminal positionsĢ. Sum the valence electrons from all the atoms.ģ. Use a pair of electrons to form a bond between each pair of bound atoms.Ĥ. Add the remaining electrons to satisfy the octet for more electronegative atom first.ĥ. If any atoms lack an octet, make a double or triple bond to give them an octet. * Hydrogen atoms are always terminal (only one bond) Write the correct skeletal structure for the molecule. Here are the summary steps for drawing Lewis structures, and if you need to review it, go over the article here.ġ. 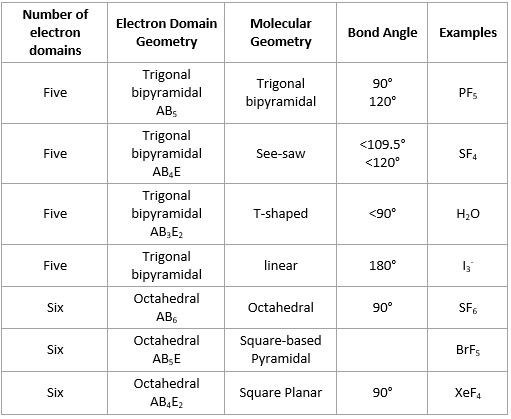
The first thing you need to do is draw the Lewis structure including all the lone pairs of electrons if there are any. This number is called the electron groups (EG) or steric number (SN) and it defines the geometry of the molecule.įor example, let’s determine the steric number of Be in BeCl 2. To determine the geometry, we count the sum of the number of atoms and lone pairs around the central atom. It is based on the principle that atoms and lone pairs repel each other because of electrostatic forces and thus, they are positioned as far away as possible to give the molecule an optimal geometry. The VSEPR (valence shell electron pair repulsion) theory predicts the geometry of atoms or molecules. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
